Neeru Bhatt
1Department of Food Science and Nutrition, P.O. Box 34, Al-Koud-123, College of Agricultural and Marine Sciences, Sultan Qaboos University, Sultanate of Oman
*Corresponding Author: Dr. Neeru Bhatt. Email: [email protected]
DOI: http://dx.doi.org/10.14206/canad.j.clin.nutr.2013.02.05
Abstract
Conjugated linoleic acid (CLA, 18:2) is a mixture of positional and geometric isomers of linoleic acid with two conjugated double bonds at various carbon positions in the fatty acid, found in nature, and do not have any known negative side effects. The most abundant source of natural CLA is the meat and dairy products of grass-fed ruminants such as cows, sheep and goats. CLA is synthesized endogenously by two pathways. The beneficial effects of CLA is reducing body fat accretion and increasing the percentage of body protein, delaying the onset of type II diabetes, retarding the development of atherosclerosis, improving the mineralization of bone, anticarcinogenic. CLA may be a more potent antioxidant than α-tocopherol and almost as effective as butylated hydroxytoluene (BHT). Various animal and human studies have shown that CLA can modulate both innate and adaptive immunity.
Full Text
INTRODUCTION
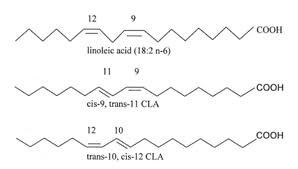
Conjugated linoleic acid is a fatty acid found in nature, and does not have any known negative side effects. CLA was known from 1930 but it was isolated in 1987 as an antimutagenic agent by Dr Pariza and opened door for new research in the arena of fatty acids. Conjugated linoleic acid (CLA, 18:2) is a mixture of positional and geometric isomers of linoleic acid with two conjugated double bonds at various carbon positions in the fatty acid (FA) chain (figure 1) such as (7,9), (8,10) (9,11]), (10,12), and (11,13). Each double bond can be either cis or trans, but those with one trans double bond are the bioactive isomers (1). There are 28 different isomers with slightly different from each other with their chemical bonds; but cis-9, trans-11 and trans-10, cis-12 is the most studied isomers (2). The double bonds positioned between the 9th and 11th carbon atoms as cis 9, trans-11 is referred rumenic acid on the basis of its ruminant origin (2).
The most abundant source of natural CLA is the meat and dairy products of grass-fed ruminants such as cows, sheep and goats. The average CLA content in meat products of ruminant and non-ruminant is reported as 0.46% and 0.16% of fat by Dhiman et al (3) whereas Shantha et al,, Mir et al (4-5) reported a higher range of CLA (1.7 to 8.5 mg CLA/g lipid) in meat products of ruminants. The CLA content in meat depends on variable factors such as species, breed, feeding regimes (5-6) (and management strategies used to raise cattle (3, 7). Breeds of cattle that deposit high amounts of fat in muscle will evidently provide a higher amount of CLA in their products (7). The total CLA content of specific foods may vary widely (3) and it is relatively easy to raise the concentration of cis-9, trans-11 CLA in ruminant lipids through manipulation of animal diet (8). Grass-fed animals are rich in CLA in comparison to stall fed animals. Substantially higher concentration of CLA was found in spring and summer season (when cows were pastured) than in fall and winter (when cows were stall-fed) (9). Further research has shown that the cows that graze at relatively higher altitudes may produce the healthiest milk of all compared with lowland grazers; milk from high altitude grazers (3700-6200 ft) has even more omega-3 fatty acids and CLA and significantly less saturated fat. Because Plants growing in higher altitudes have more omega-3 fatty acids which solidify at lower temperatures than other fats and therefore acts as a form of anti-freeze. The cows graze this enriched pasture and pass the nutrients on to their milk (10).
Grazing beef steers on pasture or increasing the amount of forage (grass or legume hay) in the diet has been shown to increase the CLA content in the fat of cattle. Supplementation of high-grain diets of beef cattle with oils (e.g., soybean oil, linseed oil, and sunflower oil) may also increase the CLA content of beef, high fat diet such as soybean significantly increased CLA in cow milk (11). Beef fat from steers fed control diet or the same diet supplemented with 2% or 4% soybean oil, respectively, contained 0.1, 1.2, or 1%, of trans-10, cis-12 CLA, respectively (11). CLA in meat is stable under normal cooking and storage condition (3). Different species of animal have different amount of CLA such as horses have the lowest CLA content and sheep the highest. Human milk is in the middle and the CLA contain in milk is as mare’s milk < sow’s milk < human milk < goat’s milk < cow’s milk < ewe’s milk (12).
CLA occurs in eggs and fat of fish and sea animals as well (Table 1). Besides animal products white button Mushrooms (13) and pomegranate seed oil (14) is also reported as natural food sources of CLA. The low concentrations of CLA have also been reported in vegetable oils (Table 1) and infant formulas (15-16)
Biosynthesis of CLA
CLA is synthesized endogenously by two pathways. In the first pathways the biohydrogenation of ingested forage-derived dietary unsaturated fatty acids such as oleic acid and lenoleic acid (LA) convert to Stearic acid (17) by the rumen bacteria (18). The intermediates produced during biohydrogenation are cis -9, trans 11 (C18:2) (the main CLA isomer in milk) and trans 11 (C18:1) trans vaccenic acid. These intermediates are accumulated and absorbed in the intestine and incorporated to various tissues. The second pathway occurs in adipose tissue and in mammary gland of lactating cows by D9-desaturase (19). The trans-Δ11-vaccenic acid (t-VA), produced as a rumen biohydrogenation intermediate from both linoleic acid and α-linolenic acid and other PUFAs (20) provides a major alternate route for CLA biosynthesis in mammalian cells, including humans, viaΔ9 desaturation by stearoyl-CoA desaturase (SCD). The mammary gland and adipose tissue of ruminants have substantial D9-desaturase activity to perform the whole process (21-24). In ruminants Δ9-desaturase activity is high in adipose tissue of growing animals, mammary tissue and adipose tissue of lactating animals; mRNA and protein for this enzyme are negligible in liver. The second most prevalent CLA isomer in milk fat is trans-7, cis-9 and it originates almost exclusively from endogenous synthesis involving Δ9-desaturase and trans-7 (C18:1) produced in the rumen.
Absorption of CLA
Though some bacterial species in the large intestine of simple stomach animals can synthesize CLA (25) but the CLA formed in the large intestine does not be subsequently absorbed. After formation in the rumen cis-9, trans-11 CLA may be directly absorbed and further metabolized (biohydrogenated) by rumen microorganisms to trans-11-octadecenoic acid. Trans-11-octadecenoic acid may then be converted back to cis-9, trans-11 CLA within mammalian cells (26-27) by stearoyl–CoA desaturase (SCD). This is the major pathway in the formation of cis-9, trans-11 CLA in cow’s milk (20, 28). Cow’s milk also contains trans-10, cis-12 CLA, and trans-10-octadecenoic acid as well (20).
Health Aspect of CLA
Dietary supplementation of CLA has been effective in reducing the percentage of body fat and increasing the percentage of body protein (29). Several in-vivo studies with rodents, pigs and cattle have shown that total body fat was reduced when animals were fed on a mixture of the two CLA isomers from synthetic sources (5, 30-33), may be due to inhibition of proliferation of adipocytes by the bio-formed CLA. CLA also helps in lessening leptin a hormone associated with weight gain and fat storage (34). CLA inhibits cancer by blocking the growth and metastatic spread of tumors. CLA inhibit both malignant and benign tumors almost immediately (35). The 10-CLA isomer seems to work preferentially through modulation of apoptosis and cell cycle control, while 9-CLA isomer affects arachidonic acid metabolism (36). CLA isomers have also been shown to have variable effects on bone formation (osteosynthesis) and resorption in animals. Dietary CLA inhibits eddosteal bone resorption, increases endocortical bone formation, and modulates the action and expression of COX enzymes, thereby decreasing prostaglandin-dependent bone resorption (37-38).
Since CLA can affect inflammatory cytokines, it is hypothesized that CLA may be a good tool for prevention or reduction of rheumatoid arthritis symptoms in humans (39). CLA is a potent anti-atherogenic dietary fatty acid in animal models of atherosclerosis by activating PPARs (40-41). CLA is very helpful in diabetic management and glycemic control, especially type II diabetes. Many studies strongly suggest that the 10-CLA isomer may be the bioactive isomer of CLA to influence the body weight changes observed in subjects with type II diabetes, a mixture of CLA (9- and 10-CLAs) rather than single could be more beneficial for the management of insulin resistance (42). However human studies have not shown this effect.
CLAs can alter the growth of neoplastic cells by influencing cell replication, interfering with components of cell cycle, or increasing cell death by promoting necrosis or/and apoptosis. Necrosis generally result from insult or toxicity reaction and triggers inflammation, whereas apoptosis is a distinct energy requiring process of programmed cell death, characterized by DNA fragmentation, chromosome condensation, nuclear fragmentation, formation of apoptotic bodies, and inversion of phosphatidylserine in the plasma membrane. CLA can reduce cell proliferation by blocking DNA synthesis (43) and cell cycle proteins (31, 44) that regulate this process and may support elevated apoptosis primarily by suppressing the expression of antiapoptotic bcl-2 gene (45). With different cell lines, CLA was able to increase the IL-2 and IFN-γ via modulation of protein kinase activity and production of oxidant species, which significantly inhibited proliferation (46). Thus, CLA has potential health benefits and can be used as prevention and treatment of many pathogenic conditions, we here will emphasize only on immunomodulatory effect of CLA.
Immune System
The immune system plays a crucial role for good health and wellbeing. It protects the body from potentially harmful substances by recognizing and responding to antigens. Antigens can be both living and non-living. Living antigens are molecules (usually proteins) on the surface of cells, viruses, fungi, or bacteria. Nonliving substances are toxins, chemicals, drugs, and foreign particles (such as a splinter). The immune system recognizes and destroys substances that contain these antigens. The body cell proteins also act as an antigen and called HLA antigen. The antigens HLAs are found in large amounts on the surface of white blood cells. They help the immune system to recognize the difference between body tissue and foreign substances. There are two types of closely connected immunities present in human defense system, the innate or nonspecific and the adaptive immunity. Innate, or nonspecific, immunity is the defense system with which a person is born. It protects against all antigens through the barriers that keep harmful materials from entering in to the body. These barriers form the first line of defense in the immune response (47-48). It includes cough reflex, enzymes in tears and skin oils, mucus, which traps bacteria and small particles, skin, stomach acid and of broad pattern recognition that leads to phagocytosis and extracellular killing of invading agents. The cells of the innate immune response such as monocytes or macrophages and natural killer cells secrete cytokines (TNF-_, IL-1, and IL-6), prostaglandins, and leukotrienes early during the nonspecific immune response and are associated with inflammatory reaction. Prostaglandins have local effects and mainly perform in the tissues where they are synthesized.
Another kind of immunity is acquired or adaptive immunity develops with exposure to various antigens. Body’s immune system builds a defense that is specific to that antigen. The adaptive immune system consists of B and T lymphocytes, which elicit their effect or functions in an antigen-specific fashion. Development of effector cells is driven by the action of T helper cells, which can be divided on the basis of the cytokines they produce into Th1 (interferon-_ (IFN-), interleukin 2 (IL- 2)] and Th2 (IL-4, IL-5, IL-10) cells. After antigenic stimulation, a subset of lymphocytes become memory cells, which can elicit faster and more potent immune responses at a subsequent exposure with the same antigen (47). Lymphocytes and natural killer cells are derived from undifferentiated, self-renewing hemopoietic stem cells through highly regulated differentiation and maturation processes. These processes are mediated by micro-environmental factors, including cell-to-cell interactions, cytokines, and growth factors (48-49).
CLA Influence on Thymocyte Differentiation
CLA can influence critical pathways of thymocyte differentiation. Thermocytes are hematopoetic progenitor cells present in thymus. With regard to the effects of CLA on immune cell development, phenotypic analysis of T cell subsets in the thymus and peripheral blood revealed that CLA acted first on immature thymocytes (ie, day 35 of dietary supplementation) and later modulated mature peripheral blood T cells (i.e. days 49 to 72 of dietary supplementation) in a pig model (50) .
Innate Response of CLA
CLA in the diet of animals affect different immune-related mechanisms involved in both allergic reactions and infections (51). O’Shea et al (51) further reported that the decrease in leukotriene and prostaglandin production in organs from CLA-fed animals. Leukotrienes mediate many of the inflammatory phenomena that are characteristic of immediate hypersensitivity reactions such as direct effects on smooth muscle cells and blood vessels contributing to the recruitment of inflammatory cells. The effects of CLA on aspects of the innate response to viral infection such as increased organ weight at the sight of infection because of increased production and recruitment of inflammatory cells coupled with the reduction in viral particles in CLA-fed animals indicate a reduction in the adverse effects resulting from influenza infection.
CLA Effect on Adaptive Immunity
CLA can modulate both innate and adaptive immunity (52). It is well documented that CLA enhanced certain immune responses while reducing the adverse effects of immune-mediated catabolism (53-54). Dietary CLA has been shown to increase immunoglobulin production in rat spleen lymphocytes (55) and to reduce antigen-induced histamine and PGE2 release from sensitized guinea pig tracheae (55). The precise roles of the individual CLA isomers in these effects are not yet known, but it has been reported that trans-10, cis-12 CLA increases lymphocyte proliferation in vitro (56).
CLA enhances immune function in-vitro and in-vivo conditions. O’Shea, et al (57) investigated the potential of CLA to modulate the humoral and cell mediated immune responses in human immune system using the two main isomers in different ratios (50:50 and 80:20 of cis-9, trans-11 and trans-10, cis-12 CLA, respectively). Hepatitis B (Hbs) vaccination was used as an infection model to investigate the humoral and cell mediated immune response. Hepatitis B antibody titres were evaluated for each subject on day 0 and 2 weeks post initial vaccination and final booster. Mean serum Hbs antibody concentration at day 85 was twice as high for subjects consuming CLA 50:50 compared with the control or the 80:20 groups. The seroprotection rate (SPR, i.e. the number of subjects with anti-Hbs concentrations >10 IU/L compared to the number of subjects with titers <10 IU/L) was significantly higher (P>0.05) for the 50:50 group compared with the control or the 80:20 group.
The cell mediated immune response was measured using the CMI multi test for Delayed-Type Hypersensitivity (DTH). Evaluation of the DTH responses on 7 recall antigens, at different time points showed no statistically significant differences in all groups. Sugano et al (58) observed increased concentration of IgA, IgG, and IgM and a drop in IgE concentration in the serum of 7-week-old rats fed with 1% CLA (50:50 isomers) mix, may be due to a shift towards Th-1 cytokine profile in. A similar effect in humans after supplementation with a 50:50 CLA isomer mixture for 12 weeks was observed by Song et al (59). CLA supplementation also decreased the levels of the proinflammatory cytokines, TNF-alpha and IL-1beta (P < 0.05), but increased the levels of the anti-inflammatory cytokine, IL-10 (P < 0.05). Another aspect of immune function, delayed type hypersensitivity (DTH) response, was decreased during and after CLA supplementation (P < 0.05). However, plasma glucose, lipids, lymphocyte phenotypic results were not affected significantly by CLA (59). Hayek et al (60) observed that dietary CLA enhances in- vitro T cell function but has no effect on in-vivo T cell-mediated function as measured by the DTH skin reaction or B cell and NK activity. The immunostimulatory effect was more pronounced in young than in old mice and was not mediated through a change in PGE2 or IL-1 production.
However, Yamasaki et al (55) found no significant effects on serum IgA, IgG, or IgM concentration after feeding 5-wk-old rats a 50:50 CLA isomer mixture for 3 weeks at doses ranging from 0.05% to 0.5%. A study carried out in another species during gestation and lactation has reported an increment concentration of serum IgG (61), a fact that supports humoral enhancement effects of CLA in early age. The specific mechanism by which CLA enhances IgA levels at mucosal sites remains unknown. But since CLA has been shown to suppress IL-4 production in-vitro (61), attenuate Th2 responses in challenged animals and regulate the number and effectors functions of several lymphocytes (50), CLA is modulating the effects of TGFb on IgA production, it is probably due to posttranscriptional and/or translational regulation, which are important in this cytokine, because it has been suggested that TGFb mRNA levels do not completely correlate with the quantity of protein produced (62). On the other hand, the increase of IgA as result of CLA supplementation might be independent of the isotype switching mechanism produced by TGFb, which has been described, but is not completely defined (63).
Immune Mechanism of CLA
The immunological effects of CLA are not well known. Several studies have suggested that it may be due to the antioxidant properties of CLA, which were demonstrated in cell free systems (64), liver microsomes (65) and in mammary glands (66). It was suggested that CLA may be a more potent antioxidant than α-tocopherol and almost as effective as butylated hydroxytoluene (BHT) (64, 67).
Two main hypotheses to explain the immuno-enhancing effects of dietary CLA have been proposed. The first one is that CLA interact with peroxisome proliferators activated receptors (PARS). PPARs (α, β, γ and δ) are nuclear receptors that translate nutritional and /or pharmacological stimuli into changes in gene expression (68) and it has a pivotal role in the stimulation of IgA responses at mucosal sites (69).The specific mechanism by which CLA enhances IgA levels at mucosal sites is unknown, but since CLA has been shown suppress IL-4 production in-vitro (70), attenuate Th2 responses in challenged animals (71) and regulates the number and effectors functions of several lymphocyts (50). PPARs bind to PPAR receptors and suppress or induce the transcription of target genes. The change in gene expression shows effect in myriad of cellular metabolic pathways such as lipid, carbohydrate and energy metabolism in immune and non-immune cells (72). The CLA isomers are active modulators of PPARs (73-74). These receptors enhance immune response and regulate gene expression. The synthetic PPAR-γ agonist inhibits the proinflammatory cytokines affecting the differentiation of monocytes and macrophages (75).
PPAR-γ comprises two isoforms, PPAR-γ 1 and PPAR- γ 2. Both are expressed in adipocytes, but PPAR- γ 1 is expressed in T and B cells, monocytes, dendritic cells, and epithelial cells (76). There are several possible options through which CLA might act. Ponferrada et al (77) reported that PPAR-γ agonists can revert stress-induced decrease of IgA production in the colon mucosa, even beyond the IgA controlled basal concentration. Infect PPAR- γ acts through modulation of transcriptional factors such as NF-kB, AP1, and STAT1 (78), which are involved in B-cell regulatory processes.A close links between intestinal-microbial interactions and regulation of PPAR-γ expression by epithelial cells of colon tissue (79). CLA may be modulating the entry of luminal antigen, the capacity for direct antigenic presentation, or even the transmission of antigen to dendritic cells from the intestinal mucosa. These hypotheses are supported by the fact that dendritic cell immunogenicity is regulated by PPAR-γ CLA dietary supplementation with CLA increases the intestinal immune defenses of Wistar rats during the first stages of life (suckling and extended to early infancy) (61). CLA-dependent enhancement of humoral mucosal immune response was demonstrated by the striking increase of intestinal IgA expression in 28-day-old rats fed CLA for 4 weeks during early life. The effects of CLA are more pronounced the earlier and more long-lasting CLA dietary supplementation.
Secondly CLA may modify mediators of immunity such as eicosanoids, prostaglandins, cytokines, and immunoglobulins (51, 80-81). CLA may act in part by competing with linoleic acid in the biosynthesis of arachidonic acid (82). Cook et al (52) showed that feeding chicks a diet containing 0.5% CLA significantly reduced the level of arachidonic acid. Belury and Kempa-Steczko (83) also demonstrated that feeding rats 0.5, 1.0 and 1.5% CLA resulted in decreased arachidonic acid levels Arachidonic acid is the precursor for PGE2; thus, increased CLA intake may decrease PGE2 production and suppressive effect on IL-2 production and T cell proliferation (68, 84). In- vitro and in- vivo studies in various animal models demonstrate that CLA influences cytokine and prostaglandin production, which could influence the inflammatory response (80).
In experimental animals, CLA has been shown to modulate the immune system and prevent immune-induced wasting (51) as well as cancer-induced wasting (85). Some studies in humans suggest a potentially beneficial effect of CLA on immune function (81). A randomized controlled trial in 28 healthy adults showed that 3g/day of CLA (mixed isomer) fed for 12 weeks, improved cell mediated immunity which allows the body to resist viruses, bacteria, fungi, and tumors (59). Improvements in other markers of immune function with CLA supplementation indicated that CLA was capable of reducing inflammatory responses as well as allergic reactions (59). A primary mechanism for immune-modulation is the multiple antioxidant capability which can reduce the deleterious effects of reactive oxygen species and free radicals, leads to premature death of immune cells (86).CLA can enhance intracellular enzyme concentration and consequently to enhance immune system.
CLA also has the ability to increase total GSH (GSH+GSSG) amount and gamma-glutamylcysteine ligase (gammaGCL) protein expression and is associated with the inhibition of typical pathological signs in mice. Glutathione (GSH) is often referred to as the body’s master antioxidant, synthesized endogenously all throughout the body and can be found in virtually every cell of the human body. Glutathione is also important in detoxification of electrophilic xenobiotics, modulation of redox regulated signal transduction, storage and transport of cysteine, regulation of cell proliferation, synthesis of deoxyribonucleotide synthesis, regulation of immune responses, and regulation of leukotriene and prostaglandin metabolism (87). Bergamo, et al (88) studied the effect of CLA on antioxidant status of pregnant mice. Significantly higher total GSH and Trolox equivalent antioxidant capacity (TEAC) levels were measured in serum of CLA-treated dams (and their pups), as compared with controls. GSH homeostasis plays a key role in the treatment of diseases in which cytokines are major participants in their pathophysiology (89).
References
- Jensen RG. The composition of bovine milk lipid: January 1995 to December 2000. J. Dairy Sci. 2002, 85:295-50
- Kramer, JKG, Sehat N, Dugan MER, Mossoba MM, Yurawecz MP, Roach JAG. et al. Distributions of conjugated linoleic acid (CLA) isomers in tissue lipid classes of pigs fed a commercial CLA mixture determined by gas chromatography and silver ion-high performance liquid chromatography. Lipids. 1998, 33: 549-58.
- Dhiman TR, Nam SH, Ure AL. Factors affecting conjugated linoleic acid content in milk and meat. Crit. Rev. Food Sci. Nutr. 2005. 45(6): 463-82.
- Shantha NC, Crum AD, Decker EA: Evaluation of conjugated linoleic acid concentrations in cooked beef. J Agric Food Chem.1994, 42:1757-60.
- Mir PS, Okine EK, Goonewardene L, He ML, Mir Z. Effects of synthetic conjugated linoleic acid (CLA) or bio-formed CLA as high CLA beef on rat growth and adipose tissue development. Can J Anim Sci.2003, 83:583-92.
- Mir PS, Mir Z, Kuber PS, Gaskins CT, Martin EL, Dodson MV, Elias Calles JA, Johnson KA, Busboom JR, Wood AJ, Pittengers GJ, Reeves JJ: Growth, carcass characteristics, muscle conjugated linoleic acid (CLA) content and plasma insulin concentrations in response to intravenous glucose challenge in high percentage Wagyu, Wagyu × Limousin and Limousin steers fed sunflower oil containing diets. J Anim Sci. 2002, 80:2996-300
- Steinhart H., Rickert R., Winkler K. Identification and analysis of conjugated linoleic acid isomers (CLA). Eur J Med Res. 2003, 8(8): 370-72.
- Khanal RC and Olson KC. Factors affecting conjugated linoleic acid (CLA) content in milk, meat, and egg: a review. Pak J Nut. 2004, 3(2):82-98.
- Parodi PW. Conjugated linoleic acid: The early years. In: Yurawecz MP, Mossoba MM, Kramer JKG, Pariza MW, Nelson G, editors. Advances in conjugated linoleic acid research, vol. 1. Champaign: AOCS Press, 1999, pp. 1–11.
- Hauswirth CB, Scheeder MR, and. Beer, JH. High Omega-3 Fatty Acid Content in Alpine Cheese: The Basis for an Alpine Paradox. Circulation. 2004, 109 (1): 103-7
- Dhiman TR, Olson KC, MacQueen IS, Pariza MW. J Dairy Sci 1999; 82(Suppl. 1):84.
- Jahreis, G. The potential anticarcinogenic conjugated linoleic acid in milk of different species: cow, goat, ewe, sow, mare, woman. Nutr Res. 1999. 19:1541-9.
- Chen S, Oh SR, Phung S. Anti-aromatase activity of phytochemicals in white button mushrooms (Agaricus bisporus). Cancer Res.2004; 66(24): 12026-34.
- Tsuzuki T, Kawakami Y, Abe R. Conjugated linolenic acid is slowly absorbed in rat intestine, but quickly converted to conjugated linoleic acid. J. Nutr. 2006; 136: 2153-59.
- Lin H, Boylston TD, Chang MJ, Luedecke LO, Shultz TD. Survey of the conjugated linoleic acid content of dairy products. J. Dairy Sci.1995. 78:2358-65.
- Fritsche J, Steinhart H. Amounts of conjugated linoleic acid (CLA) in German foods and evaluation of daily intake. Z. Lebensm. Unters. Forsch. A, 1998, 206:77-82.
- Griinari JM, Chouinard PY, Bauman DE. Tran fatty acid hypothesis of milk fat depression revised. In: Proc. Cornell Nutr. Conf. Feed Manuf. Cornell University, Ithaca, NY 1998 pp. 208–216.
- Harfoot CG and Hazlewood GP. Lipid metabolism in the rumen. In: The Rumen Microbial Ecosystem (Hobson, P. N., ed.), pp. 285–322, Elsevier Applied Science Publishers, London, UK. 1988.
- Griinari JM, Cor BA, Lacy SH, Chouinard PY, Nurmela KVV and Bauman DE. Conjugated Linoleic Acid Is Synthesized Endogenously in Lactating Dairy Cows by D9-Desaturase. J. Nutr. 2000; 130: 2285–91
- Grignard JM, Bauman DE. Biosynthesis of conjugated linoleic acid and its incorporation into meat and milk ruminants. In: Yurawecz MP, Mossoba MM, Kramer JKG, Pariza MW, Nelson G, editors. Advances in Conjugated Linoleic Acid Research, Volume 1. Champaign: AOCS Press. 1999. pp180–200.
- Kinsella JE. Stearyl CoA as a precursor of oleic acid and glycerolipids in mammary microsomes from lactating bovine: possible regulatory step in milk triglyceride synthesis. Lipids. 1972, 7: 349–55
- Martin GS, Lunt DK, Britain KG, Smith SB. Postnatal development of stearoyl coenzyme A desaturase gene expression and adiposity in bovine subcutaneous adipose tissue. J. Anim. Sci. 1999, 77: 630–36.
- St. John LC, Lunt DK, Smith SB. Fatty acid elongation and desaturation enzyme activities of bovine liver and subcutaneous adipose tissue microsomes. J Anim Sci. 1991, 69: 1064–73.
- Ward RJ, Travers MT, Richards SE, Vernon RG, Salter AM, Buttery PJ, Barber MC. Stearoyl-CoA desaturase mRNA is transcribed from a single gene in the ovine genome. Biochim. Biophys. Acta, 1998, 1391: 145–56.
- Chin SF, Storkson JM, Liu W, Albright KJ, Pariza MW: Conjugated linoleic acid (9, 11- and 10, 12-octadecadienoic acid) is produced in conventional but not germ-free rats fed linoleic acid. J Nutr. 1994a. 124:694–701,
- Holman RT, Mahfouz MM. Cis and trans octadeconoic acid as precursor of polyunsaturated fatty acids. Prog Lipid Res.1980, 20:151–6.
- Pollard MR, Gunstone FD, James AT and Morris LJ. Desaturation of positional and geometric isomers of monoenoic fatty acids by microsomal preparations from fat liver. Lipids, 1980, 15: 306-14
- Santora JE, Palmquist DL, Roehrig KL. Trans-vaccenic acid is desaturated to conjugated linoleic acid in mice. J Nutr. 2000, 130: 208–15.
- Syvertsen C, Halse J, Hoivik HO, Gaullier JM, Nurminiemi M, Kristiansen K, Einerhand A, O’Shea M, Gudmundsen O: The effect of 6 months supplementation with conjugated linoleic acid on insulin resistance in overweight and obese. Internat J Obesity 2007, 31:1148-54.
- Park Y, Albright KJ, Liu W, Storkson JM, Cook ME, Pariza MW: Effectof conjugated linoleic acid on body composition in mice. Lipids. 1997, 32:853-58.
- Dugan MER, Aalhus JL, Schaefer AL, Kramer JKG. The effect of conjugated linoleic acid on fat to lean repartitioning and feed conversion in pigs. Can J Anim Sci. 1997; 77: 723–5.
- Szymczyk B, Pisulewski P, Szczurek W, Hanczakowski P: The effect of feeding conjugated linoleic acid (CLA) on rat growth performance, serum lipoproteins and subsequent lipid composition of selected rat tissues. J Sci Food Agric. 2000; 80: 1553-58.
- Poulos SP, Sisk M, Hausman DB, Azain MJ, Hausman GJ: Pre- and Postnatal dietary conjugated linoleic acid alters adipose development, body weight gain and body composition in Sprague-Dawley rats. J Nutr.2001; 131:2722-31.
- Belury MA, Mahan A and Banni S. The conjugated linoleic acid (CLA) isomer, t10c12-CLA, is inversely associated with changes in body weight and serum leptin in subjects with type 2 diabetes mellitus J Nutr.2003; 133(1): 257S-60S
- Belury MA, Moya-Camarena SY, Lu M, Shi L, Leesnitzer LM, Blanchard SG: Conjugated linoleic acid is an activator and ligand for peroxisome proliferator-activated receptor-gamma (PPAR).Nutr Res. 2002; 22:817-24.
- Ochoa JJ, Farquharson AJ, Grant I, Moffat LE, Heys SD, Wahle KW: Conjugated linoleic acids (CLAs) decrease prostate cancer cell proliferation: different molecular mechanisms for cis-9, trans-11 and trans-10, cis-12 isomers. Carcinogenesis 2004; 25:1185-91.
- Watkins BA, Li Y, Lippman HE, Seifert MF: A test of Ockham’s razor: implications of conjugated linoleic acid in bone biology.Am J Clin Nutr 2004, 79:1175S-85S.
- Tarnopolsky MA, Safdar A: The potential benefits of creatine and conjugated linoleic acid as adjuncts to resistance training in older adults. Appl Physiol Nutr Metab. 2008; 33:213-27
- Kelly O and Cashman KD. The effect of conjugated linoleic acid on calcium absorption and bone metabolism and composition in adult ovariectomised rats. Prostag Leukotr Essential Fatty Acids. 2004; 71:295-301
- Weldon S, Mitchell S, Kelleher D, Gibney MJ, Roche HM: Conjugated linoleic acid and atherosclerosis: no effect on molecular markers of cholesterol homeostasis in THP-1 macrophages. Atherosclerosis. 2004; 174:261-73.
- Naumann E, Carpentier YA, Saebo A, Lassel TS, Chardigny JM, Sebedio JL, Mensink RP: Cis-9, trans-11 and trans-10, cis-12 conjugated linoleic acid (CLA) do not affect the plasma lipoprotein profile in moderately overweight subjects with LDL phenotype B. Atherosclerosis. 2006; 188:167-74.
- Taylor CG, Zahradka P: Dietary conjugated linoleic acid and insulin sensitivity and resistance in rodent models. Am J Clin Nutr. 2004; 79:1164S-68S.
- Oh YS, Lee HS, Cho HJ, Lee SG, Jung KC, Park JH. Conjugated linoleic acid inhibits DNA synthesis and induces apoptosis in TSU-Pr1 human bladder cancer cells. Anticancer Res. 2003; 23:4765-72.
- Ip C, Dong Y, Ip MM, Banni S, Carta G, Angioni E et al. Conjugated linoleic acid isomers and mammary cancer prevention. Nutr Cancer. 2002; 43:52-8.
- Miller A, Stanton C, Devery R: Modulation of arachidonic acid distribution by conjugated linoleic acid isomers and linoleic acid in MCF-7 and SW480 cancer cells. Lipids 2001
- Luongo D, Bergamo P, Rossi M: Effects of conjugated linoleic acid on growth and cytokine expression in Jurkat T cells. Immunol Lett. 2003; 90:195-201.
- Griffiths M. Nonspecific protective mechanisms. Introduction to human physiology. 2nd ed. New York: Macmillan Publishing. pp331–53. 1981
- Firestein GS. The inflammatory response. In: Goldman L, Ausiello D, eds. Cecil Medicine. 23rd ed. Philadelphia, Pa: Saunders Elsevier. 2007. Chap 45.
- Goronzy JJ, Weyand CM. The innate and adaptive immune systems. In: Goldman L, Ausiello D, eds. Cecil Medicine. 23rd ed. Philadelphia, Pa: Saunders Elsevier.2007chap 42.
- Bassaganya-Riera J, Hontecillas R, Zimmerman DR, Wannemuehler MJ. Dietary conjugated linoleic acid modulates phenotype and effector functions of porcine cd8 (+) lymphocytes. J Nutr. 2001; 131: 2370–7.
- O’Shea, M, Bassaganya-Riera, J and Mohede, ICM. 2004. Immunomodulatory properties of conjugated linoleic acid. Am J Clin Nutr 2004; 79(suppl):1199S–206S
- Pariza MW, Park Y and Cook ME. Mechanisms of action of conjugated linoleic acid: evidence and speculation. Proc. Soc. Exp.Biol. Med. 2000; 223: 8–13.
- Cook ME, Miller CC, Park Y, Pariza M. Immune modulation by altered nutrient metabolism control of immune-induced growth depression. Poultry Sci. 1993; 72: 1301–5
- Miller CC, Park Y, Pariza MW, Cook ME. Feeding conjugated linoleic acid to animals partially overcomes catabolic responses due to endotoxin injection. Biochem Biophys Res Commun. 1994; 198:1107–12.
- Yamasaki M, Kishihara K, Mansho K, Ogino Y, Kasai M, Sugano M et al. Dietary conjugated linoleic acid increases immunoglobulin productivity of Sprague-Dawley rat spleen lymphocytes. Biosci Biotechnol Biochem. 2000; 64: 2159–64
- DeLany JP, Pariza MW, Cook ME. Conjugated linoleic acid rapidly reduces body fat content in mice without affecting energy intake. Am J Physiol. 1999; 276(4):1172-9.
- O’ Shea, M, Albers R, van der Wielen R, Brink L, Dorovska-Taran, V, Mohede. Seroprotection. CLA Stimulates Antigen Specific Antibody Production in Humans. Conjugated Linoleic Acid Research, Current Status and Future Directions May 15-16, 2002 Lister Hill Auditorium Bethesda, Maryland
- Sugano M, Tsujita A, Yamasaki M, Noguchi M, Yamada K. Conjugated linoleic acid modulates tissue levels of chemical mediators and immunoglobulins in rats. Lipids. 1998; 33:521–7.
- Song HJ, Grant I, Rotondo D, Mohede I, Sattar N, Heys SD, Whale KW. Effect of CLA supplementation on immune function in young healthy volunteers. Eur J Clin Nutr. 2005; 59(4): 508-17.
- Hayek MJ, Nim Han S, Wu D, Watkins A, Meydani M, Dorsey JL, Smith DE, Meydani SN. Dietary Conjugated Linoleic Acid Influences the Immune Response of Young and Old C57BL/6NCrlBR Mice. J. Nutr. 1999; 129: 32–8.
- Pérez-Cano FJ, Ramírez-Santana C, Molero-Luís M, Castell M, Rivero M, Castellote C, Franch. A. Mucosal IgA increase in rats by continuous CLA feeding during suckling and early infancy.J. Lipid Res. 2009; 50: 467–76.
- Kim PH, and Kagnoff, MF. Effects of transforming growth factor beta-1 and interleukin-5 on IgA isotype switching at the clonal level. J. Immunol. 1990; 145: 3773–78.
- Tokuyama H and Tokuyama. Y. The regulatory effects of all-trans-retinoic acid on isotype switching: retinoic acid induces IgA switch rearrangement in cooperation with IL-5 and inhibits IgG1 switching. Cell. Immunol. 1999; 192: 41–47
- Ha YL, Storkson J, Pariza MW. Inhibition of benzo(a) pyreneinduced mouse forestomach neoplasia by conjugated dienoic derivatives of linoleic acid. Cancer Res. 1990; 50: 1097–101.
- Pariza MW, Ha YL, Benjamin H, Sword T, Gruter A, Chin SF, Storkson J, Faith N, Albright K. Formation and action of anticarcinogenic fatty acids in nutritional and toxicological consequences of food processing. In: (Friedman, M., ed.), pp. 269–272. Plenum Press, New York, NY.1991.
- Ip C, Chin SF, Scimeca JA, Pariza MA. Mammary cancer prevention by conjugated derivatives of linoleic acid. Cancer Res. 1991; 51: 6118–24.
- Goodwin JS, Bankhurst AD, Messner R.P. Suppression of human T cell mitogenesis by prostaglandins. J. Exp. Med. 1977; 146: 1719–34.
- Schoonjans KG, Martin BS, Auwerx J. Peroxisome proliferator-activated receptors, orphans with ligands and functions. Curr. Opin. Lipidol. 1997; 8: 159-66
- Borsutzky SBB, Cazac JR, Guzman CA. TGF-beta receptor singnaling is critical for mucosal IgA responses. J Immunology.2004; 173: 3305-9
- Cook ME. Conjugated linoleic acids role in immunity and immune related disorders. Perspectives on Conjugated Linoleic Acid Research: Current Status and Future Directions, NIH,Bethesda, MD.2002.
- Hontecillas R, Wannemeulher MJ, Zimmerman DR, et al. Nutritional regulation of porcine bacterial-induced colitis by conjugated linoleic acid. J Nutr 2002; 132: 2019–27.
- Banni, S, Angioni E, Carta G, Casu V, Deiena M, Dessi, MA. et al. Corongiu FP. Influence of dietary conjugated linoleic acid on lipid metabolism in relation to its anticarcinogenic activity. In: (Ed. M. P. Yurawecz, M. M. Mossoba, J. K. G. Kramer, M. W. Pariza and G. J. Nelson). Advances in Conjugated Linoleic Acid Research. Vol. I. AOCS Press, Champaign, IL, pp. 307-318. 1999.
- Moya-Carnarena, SY, Vanden Huevel, JP, Blanchard, SG, Leesnitzer, LA, Belury, MA. Conjugated linileic acid is a potent naturally occurring ligand and activator of PPAr-α. J Lipid Res.1999; 40: 1426-33
- Houseknecht Kl, Vanden Heuvei JP, Moya-Carnarena, SY, Protocerrero CP, Peck LW, Nickel KP. et al. Dietary conjugated linoleic acid normalizes impaired glucose tolerance in in the Zucker diabetic fatty fa/rat. Biochem Biophys Res Commun. 1998, 244: 678-82.
- Tontonoz P, Nagy L, Alverez JGA, Thomazy VA, Evans RM. PPAR-γpromotes monocytes/macrophage differentiation and uptake of oxidized LDL. Cell. 1999, 93: 241-52
- Hontecillas R and Bassaganya-Riera J. Differential requirements for proliferation of CD41 and gammadelta1 T cells to spirochetal antigens. Cell. Immunol.2003; 224: 38–46.
- Ponferrada, A, Caso JR, Alou L, Colon A, Sevillano D, .Moro MA, Lizasoain I, Menchen P, Gomez-Lus ML, Lorenzo, P. The role of PPARgamma on restoration of colonic homeostasis after experimental stress-induced inflammation and dysfunction.Gastroenterology. 2007; 132: 1791–803.
- Li Y, Watkins BA. Conjugated linoleic acids alter bone fatty acid composition and reduce ex vivo prostaglandin E-2 biosynthesis in rats fed n_6 or n_3 fatty acids. Lipids 1998; 33:417–25.
- Kelly D, Campbell JI, King TP, Grant G, Jansson EA, Coutts AG. et al. Commensal anaerobic gut bacteria attenuate inflammation by regulating nuclear cytoplasmic shuttling of PPAR-gamma and RelA. Nat. Immunol. 2004; 5:104–12.
- Bhattacharya A, Banu J, Rahman M. Biological effects of conjugated linoleic acid in health and disease. J. Nutr.Biochem.. 2006; 17: 789-10.
- Tricon S, Burdge GC, Kew S. Effects of cis-9, trans-11 and trans-10, cis-12 conjugated linoleic acid on immune cell function in healthy humans. Am. J. Clin. Nutr. 2004; 80(6): 1626-33.
- Cook ME, Miller CC, Park Y and Pariza MW. Immune modulation by altered nutrient metabolism: Nutritional control of immune-induced growth depression. Poultry Sci. 1993; 72: 1301-5.
- Belury MA, Kempa-Steczko A. Conjugated linoleic acid modulates hepatic lipid composition in mice. Lipids. 1997; 32: 199–04
- Goodwin JS, Webb DA. Regulation of the immune response by prostaglandins. Clin. Immunol. Immunopathol.1980; 15: 106–22.
- Graves E, Hitt A, Pariza MW, Cook ME, McCarthy, DEO. Conjugated linoleic acid preserves gastrocnemius muscle mass in mice bearing the colon-26 adenocarcinoma. Res. Nurs. Health. 2005; 28:48-55.
- Brennan LA, Morris GM, Wasson GR, Hannigan BM, Barnett, YA. The Effect of Vitamin C or Vitamin E Supplementation on Basal and H2O2-Induced DNA Damage in Human Lymphocytes. British Journal of Nutrition. 2000; 84: 195–202
- Sen CK: Glutathione homeostasis in response to exercise training and nutritional supplements. Molecular and Cellular Biochemistry. 1999; 196:31-42.
- Bergamo P, Maurano F and Rossi, M. Phase 2 enzyme induction by conjugated linoleic acid improves lupus-associated oxidative stress.Free Radic Biol Med.2007; 43(1):71-9.
- Haddad JJ and Harb HL. L-g-Glutamyl-L-cysteinyl-glycine (glutathione; GSH) and GSH-related enzymes in the regulation of pro- and anti-inflammatory cytokines: a signaling transcriptional scenario for redox(y) immunologic sensors(s)? Mol. Immunol. 2005; 42:987–1014.
Figure 1. Structure of Conjugated Linoleic Acid (CLA)
Table 1: CLA Content in Different Foods
|
Dairy Products |
CLA amount (mg/g) |
|
Homogenized milk |
5.5 |
|
Condensed milk |
7.0 |
|
Butter |
4.7 |
|
Butter fat |
6.1 |
|
Ice-cream |
3.6 |
|
Frozen yogurt |
2.8 |
|
Plain yogurt |
4.8 |
|
Low fat yogurt |
4.4 |
|
Medium cheddar |
4.1 |
|
Cottage cheese |
4.5 |
|
Mozzarella cheese |
4.9 |
|
Processed cheese |
5.0 |
|
Egg, meat and fish |
CLA amount (mg/g) |
|
Lamb meat |
5.8 |
|
Ground beef |
4.3 |
|
Beef |
3.8 |
|
Veal |
2.7 |
|
Salmon fish |
0.3 |
|
Lake trout fish |
0.5 |
|
Shrimp |
0.6 |
|
Chicken |
0.9 |
|
Turkey |
2.6 |
|
Egg yolk |
0.6 |
|
Plant Sources |
CLA amount (mg/g) |
|
Safflower oil |
0.7 |
|
Sunflower oil |
0.4 |
|
Corn oil |
0.2 |